|
In a simple orbit such as the Earth going about the Sun, each period is exactly the same length. However, there is a difference with the Periodic Table. After you add just enough electrons to fill up a \(p\) orbital, the next electron you add is as if you were starting over on a new shell. On the periodic table, it’s element numbers that are periodic. The period of the Earth’s orbit around the Sun is about 365 days after that much time, the Earth is back where it started. When something is periodic, it means that it has regular cycles. The name “Periodic Table” is not arbitrary. Each row on the periodic table is called a “Period”. Thus, each column on the periodic table is called a “Group”. Additionally, to fill out the current modern periodic table, we’d need to extend this to include the \(5f, 6s, 6p, 7s\), and \(7p\) orbitals.īecause the chemical properties of an element are approximately determined by how far away it is from a noble gas, constructing the periodic table in this manner means that every column should have somewhat similar chemical properties. Although not drawn, there are 14 states available in the \(4f\) orbital. States higher in the diagram are (usually!) states at higher energy, although the exact spacing on the diagram should not be interpreted as meaningful. 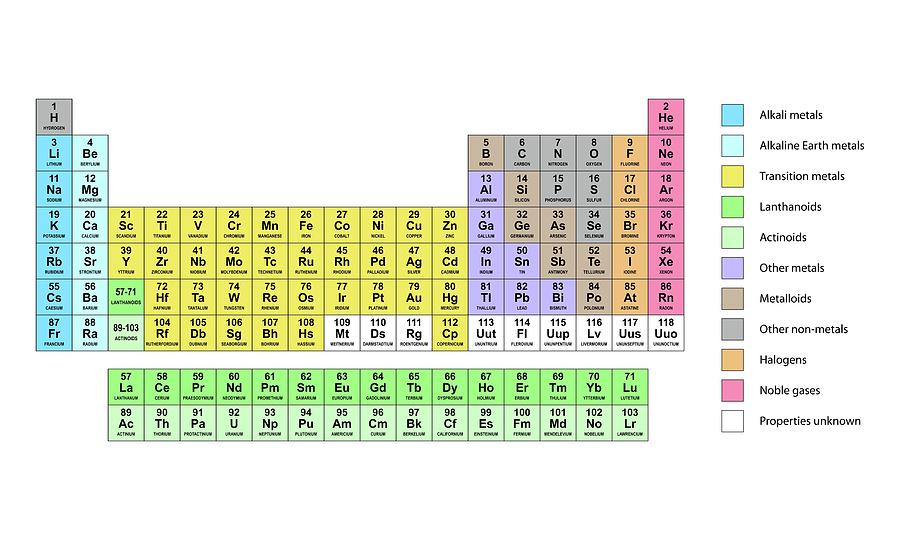 
If you completely fill a \(p\) orbital, then you’re on the very right side of the table, and the element you’ve constructed is a noble gas. As you fill in more and more levels, you move farther and farther to the right on the table. If you have just a single spare electron in an \(s\) shell, then that is an element like Hydrogen, Lithium, or Sodium it goes on the left column of the periodic table. Keep filling them in until you have as many electrons as you need. Start with the lowest energy states, and fill in available states with electrons. You can use the diagram below to figure out where an element will fall on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
